
Operating Room Doors: Key Requirements for Sterile Surgical Areas
- By:Lisa
- 2026-05-15
- 29
Drawing from extensive field deployments across tertiary hospital surgical suites, one principle stands out: the modern operating room is never a static sterile environment. It operates as a highly controlled, mission-critical space that must continuously manage dense clinical traffic, heavy equipment movement, dynamic airflow regulation, and strict infection prevention protocols.
Within this tightly coordinated ecosystem, operating room doors are frequently treated as standard architectural components. In practice, they function as an active control layer—directly influencing pressure stability, pathogen containment, workflow efficiency, and long-term asset performance. For hospital facility directors, surgical suite contractors, and medical door procurement teams, understanding how these doors perform, degrade, and require maintenance in real clinical settings is essential to avoiding project delays and reducing lifecycle costs.
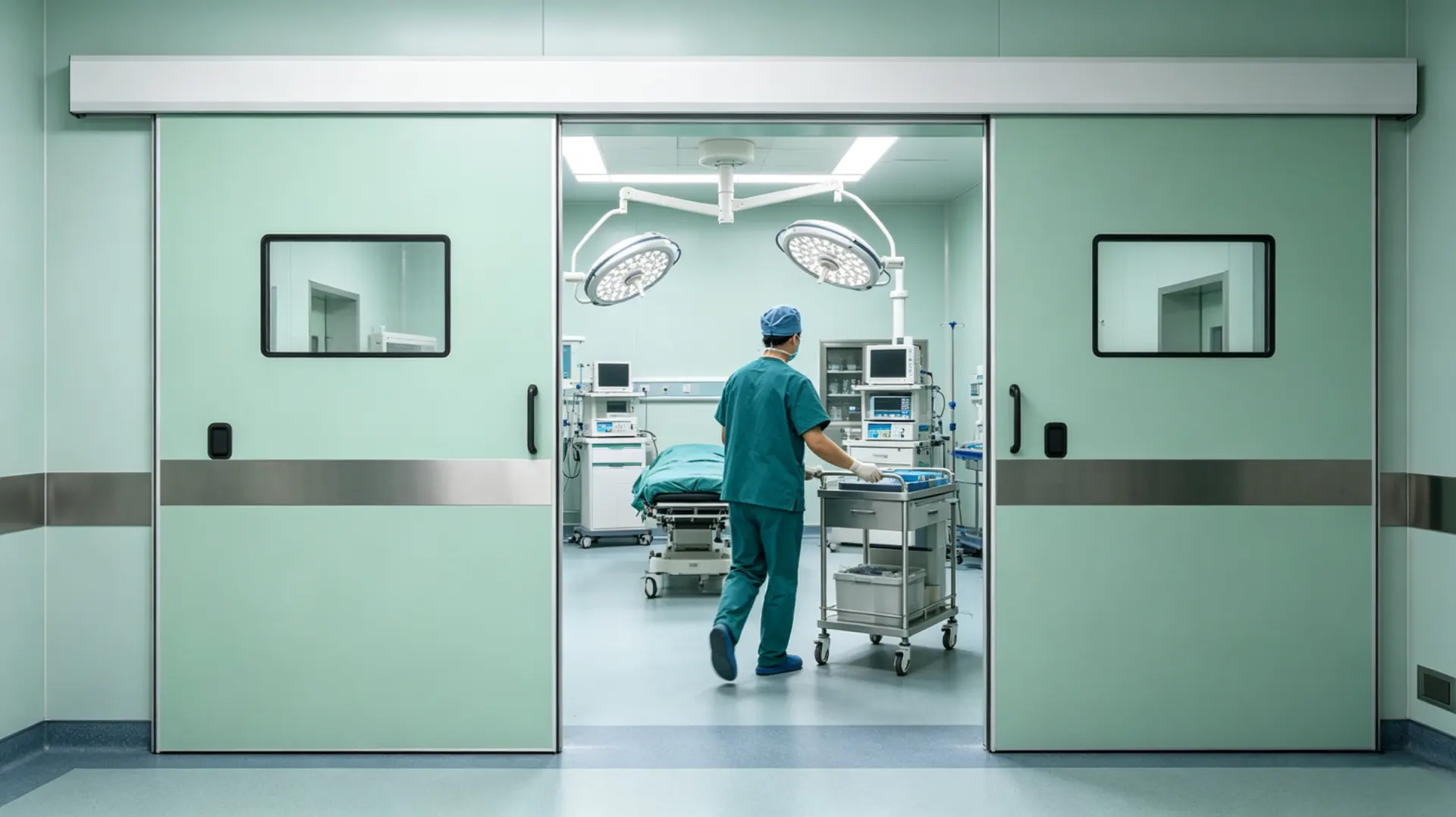
1. Infection Control: Designing for Cleanability and Pathogen Interception
Terminal cleaning protocols begin long before the first surgical case. During this process, door seams, hinges, and thresholds consistently emerge as high-risk zones for microbial accumulation. Surface-mounted hardware not only traps particulate matter but can also wick disinfectants into the door core over repeated wiping cycles, inadvertently creating harborage points.
Clinical infection control teams emphasize that infection control door systems must evolve from basic closure mechanisms to cleanable, low-retention interfaces. Field-proven design priorities include:
- Concealed hinges and integrated overhead tracks that allow EVS (Environmental Services) technicians to wipe surfaces in continuous, uninterrupted passes
- Laser-welded or seamless polymer-bonded door-to-frame joints with surface porosity maintained below 0.5μm to prevent capillary absorption of chemical agents
- Antimicrobial hospital door surfaces validated through ISO 22196 testing, demonstrating ≥99.9% reduction in Staphylococcus aureus and E. coli reproduction under real-world cleaning cycles
Disinfectant chemistries are advancing faster than material science. Hydrogen peroxide vaporization, low-temperature plasma, and quaternary ammonium blends now dominate terminal cleaning protocols. Long-term chemical compatibility directly determines whether finishes will blister, chalk, or delaminate over time. For chemical resistant medical doors, procurement teams should require documented compatibility testing against the facility's actual disinfectant SDS library.
Floor transitions demand equal precision. Threshold recesses must align with epoxy self-leveling or medical-grade homogeneous PVC flooring within a ≤1.5mm tolerance. Premature seal degradation and air leakage frequently trace back to uncoordinated floor elevations, which subject door bottoms to repeated caster impact. Impact-resistant threshold guards should be integrated to maintain seal integrity and break the contamination transfer path.
2. Airtightness & Pressure Management: Dynamic Coordination with HVAC Systems
Surgical suite positive pressure is not a fixed setpoint. It functions as a dynamic control loop: door opening instantly depressurizes the space, and the HVAC system must restore target pressure within a tightly regulated recovery window. The operational value of OR door airtightness lies in how seamlessly it integrates with building airflow management.
Per ASHRAE 170-2021 §7.3.2 and FGI Guidelines 2022, operating room positive pressure must remain stable between +2.5 Pa and +7.5 Pa. Following door closure, system recovery to the target range must occur within ≤30 seconds—commonly referenced as operating room pressure recovery.
Achieving this metric requires coordinated engineering:
- Perimeter EPDM or medical-grade silicone seals must feature auto-compensation geometry to ensure uniform contact upon closure. Compression set resistance must comply with EN 16034, limiting rebound force degradation to ≤15% after 50,000 actuation cycles
- Automatic drop-down thresholds serve as critical floor-level seals. They retract during passage and deploy upon closure to block low-level air infiltration and particulate migration
- Door actuation speed (typically 0.5–0.8 m/s) requires PID feedforward integration with VAV damper positioning and VFD fan response to prevent pressure overshoot or oscillation
- Anteroom interlock controllers must incorporate micro-pressure gradient monitoring to prevent simultaneous door openings that generate cross-drafts and compromise corridor pressurization
During commissioning, smoke visualization testing remains the most reliable field verification method. Digital manometer data should always be validated against observable airflow behavior at door perimeters.
3. Durability & Lifecycle Management: Optimizing for Total Cost of Ownership
Daily operational demands on surgical doors are extreme: continuous equipment cart traffic, C-arm maneuvering, impact from stretchers, and aggressive chemical cleaning cycles. Over a 10–15 year service life, cumulative mechanical and chemical exposure far exceeds commercial building standards.
Engineering for high-traffic surgical door systems is fundamentally an asset preservation strategy. Material selection must align with documented traffic loads:
- High-impact zones require aluminum honeycomb or high-density phenolic resin cores with flexural strength ≥120 MPa
- Surface assemblies must pass EN 160 Class 4 or ASTM F476 impact testing (150J impact without structural deformation). Internal framing should utilize ≥2.0mm galvanized steel with continuous weld construction
- Hinge assemblies rated for ≥120kg static load, equipped with sealed self-lubricating bearings, prevent sagging and binding over extended service cycles
Reactive maintenance models drive disproportionate downtime. Modern surgical door lifecycle cost strategies prioritize modular architecture: quick-release control panels, field-replaceable compression seals, and independent drive modules. Component-level replacement preserves surgical schedule continuity.
Motor duty cycles must be contractually specified as S4-40% or S5, with Class F or higher insulation ratings to withstand high-frequency thermal cycling. Procurement documentation should include approved chemical compatibility matrices and explicit warranty replacement scopes. A 10-year TCO model—incorporating initial capex, preventive maintenance, spare parts inventory, and schedule disruption costs—provides a realistic basis for vendor selection.
4. Smart Automation & System Integration: Touchless Workflow Compatibility
Sterile technique protocols eliminate physical contact with environmental controls. Surgical door automation must therefore operate seamlessly within clinical workflows: automatic activation during approach, safe closure after passage, and manual override capability during any system anomaly.
Clinical activation preferences consistently favor:
- Dual-sensor arrays (microwave radar + infrared) providing 1.5–3m detection zones with ambient light filtering to minimize false triggers
- Foot-switch backups and hardwired integration with surgical tables/booms for high-sterility scenarios
- Nuisance prevention algorithms featuring adjustable 3–8 second hold-open timers, corridor blind-zone masking, and multi-pedestrian tracking to stabilize airflow patterns
Safety redundancies are non-negotiable. Power loss or controller failure must not impede egress. Manual opening force must remain ≤130N, compliant with ADA accessibility standards and EN 16005 safety requirements. Anti-pinch protection requires dual-redundancy infrared light curtains (≤30mm resolution) and capacitive edge sensors, triggering immediate reversal upon contact detection.
Door control system integration has transitioned from standalone controllers to networked building infrastructure. Controllers should support BACnet MS/TP or Modbus RTU, transmitting actuation frequency, fault diagnostics, and motor load data to facility management platforms. HL7/FHIR-enabled systems can synchronize with EMR workflows to align room status with access permissions.
IEC 60601-1 and YY 9706.102-2021 certification establishes baseline electromagnetic compatibility. Inadequate EMC shielding risks interference with patient monitoring and life-support equipment. Mature BMS hospital integration transforms surgical doors into operational data endpoints, enabling predictive maintenance, energy optimization, and compliance auditing.
5. Workflow Planning & Emergency Egress: Balancing Throughput and Life Safety
Surgical suite circulation design directly impacts clinical response times and staff ergonomics. The surgical corridor door layout must accommodate routine equipment movement while maintaining unobstructed emergency egress paths.
Zone-specific typology selection follows established clinical logistics:
- Clean corridors connecting to operating rooms prioritize automatic hermetic sliding doors to maximize clear passage and eliminate swing-radius conflicts
- Anesthesia induction and sterilization prep areas often benefit from swinging doors, which provide superior static sealing and wider clear openings for equipment staging
- Clean/soil corridor segregation requires ≥1.5m clear opening widths. Standard stretcher and C-arm maneuvering demands a minimum 150mm lateral clearance buffer on both sides
Fire and life safety compliance overrides all operational automation. When fire-rated medical doors function as smoke or fire barriers, they must meet EN 16034 or GB 12955 standards for ≥60-minute fire integrity. Electromagnetic locks must operate on fail-safe principles, hardwired to the fire alarm control panel. Within ≤3 seconds of alarm confirmation, all egress paths must unlock automatically. Emergency egress doors always supersede automated scheduling or interlock sequences.
Status indication systems reduce workflow friction. RGB LED indicators (green: clear, yellow: cleaning/interlocked, red: fault/fire) paired with low-frequency audible cues mitigate blind-corner collisions. Proximity sensors on door frames can trigger automatic deceleration during high-speed cart approaches. Digital signage displaying real-time surgical schedules and sterile status reduces unnecessary staff entries and supports throughput optimization.
6. Procurement & Commissioning: Ensuring Field Viability Beyond Specification Compliance

The most frequent challenge in hospital door procurement is the gap between theoretical specification and site execution. Projects often feature technically compliant bidding documents, only to encounter structural load limitations, floor elevation misalignment, or conduit routing conflicts during installation.
A hospital medical door specification checklist should anchor technical coordination:
- Airtightness: Leakage rate ≤0.5 m³/h·m (Test standard: EN 12207 Class 4 / ISO 14644-3)
- Ingress Protection: IP54 (standard clean zones) / IP65 (soil corridors requiring high-pressure washdown)
- Motor Duty Cycle: S4-40% or S5 (Class F+ insulation, optimized for high-frequency cycling)
- Communication Protocols: BACnet/Modbus/HL7 compatibility matrix and API documentation
Vendor qualification must extend beyond product catalogs. Prioritize manufacturers with dedicated medical door engineering capabilities and documented delivery of ≥3 tertiary hospital surgical suites within the past three years. Third-party test reports should originate from ISO/IEC 17025-accredited laboratories, covering particle retention, electrical safety, fire resistance, airtightness, and EMC. Service agreements must define localized technical support, 24-hour response SLAs, and regional spare parts inventory for seals, controllers, and drive units. Exercise caution with general architectural door manufacturers entering medical applications, as clinical workflow integration and infection control validation often require specialized expertise.
On-site installation and commissioning validate upstream planning. Door frame anchoring must align with wall substrate capacity (steel studs, CMU, or reinforced concrete), with heavy-duty assemblies requiring pre-installed structural backing. Finished floor elevation tolerance must be maintained within ±2mm.
Field coordination failures frequently manifest during threshold deployment. A recent project experienced an 11-day commissioning delay due to unaligned floor elevations across door, flooring, and low-voltage trades. Implementing medical door installation guidelines requires cross-disciplinary elevation verification before rough-in. Commissioning protocols should include:
- Manometer zero-calibration and operating room pressure recovery validation (target ≤30s)
- Full-path interlock logic verification and anti-pinch calibration (reversal at ≤150N resistance)
- Power-loss manual egress testing (≤130N opening force)
- BMS communication handshake and data packet integrity verification (≤0.1% packet loss)
Pre-procurement installation of a 1:1 field mock-up—complete with tracks, controllers, and seals—provides irreplaceable validation. Equipment cart routing, EVS cleaning cycles, and pressure monitoring should be tested under simulated conditions. Parameters verified in the mock-up phase perform consistently across full suite deployment. The rigor of OR door commissioning directly correlates with long-term operational reliability.
Conclusion
A properly engineered surgical door operates invisibly during routine clinical workflows. System failure, however, immediately impacts surgical scheduling, infection control compliance, and staff safety. Operating room doors have evolved from passive architectural partitions into integrated control nodes managing pressurization, pathogen containment, electromechanical reliability, and clinical automation.
As AI-driven flow optimization, self-healing antimicrobial surfaces, and modular retrofit methodologies advance, these systems will continue to assume greater operational responsibility. For project owners and engineering teams, early alignment between infection control, mechanical, clinical, and architectural stakeholders—grounded in real-world workflow validation—delivers measurable value over specification compliance alone. Every door in a surgical suite ultimately guards the continuity of critical care delivery.
Note: Technical parameters and compliance frameworks referenced in this article align with current international and regional standards. Project-specific implementation requires validation against local fire codes, accreditation requirements, and institutional clinical protocols.
-
 Cleanroom Glass Windows Are The Key to Maintaining a Clean Environment
Cleanroom Glass Windows Are The Key to Maintaining a Clean Environment -
 Top Aluminium Profile Manufacturers in China: Leading the Global Market
Top Aluminium Profile Manufacturers in China: Leading the Global Market -
 The Evolution of Air Tight Sliding Doors
The Evolution of Air Tight Sliding Doors -
 AHU Aluminium Profile: A Comprehensive Guide
AHU Aluminium Profile: A Comprehensive Guide -
 The Importance of Choosing the Right Cleanroom Door in Vietnam
The Importance of Choosing the Right Cleanroom Door in Vietnam -
 The Benefits of Hospital Automatic Doors: Enhancing Efficiency and Safety
The Benefits of Hospital Automatic Doors: Enhancing Efficiency and Safety -
.jpg) The Best Bathroom Door Manufacturers - Unlocking Endless Possibilities!
The Best Bathroom Door Manufacturers - Unlocking Endless Possibilities! -
 Unlock the Possibilities with AJ Manufacturing Doors
Unlock the Possibilities with AJ Manufacturing Doors -
 Make a Statement with Manufactured Home Interior Doors!
Make a Statement with Manufactured Home Interior Doors! -
 what is aluminum profile? Aluminum Profiles for Your Home is the best option
what is aluminum profile? Aluminum Profiles for Your Home is the best option
-
 Operating Room Doors: Key Requirements for Sterile Surgical Areas
Operating Room Doors: Key Requirements for Sterile Surgical Areas -
 Isolation Room Doors: Pressure, Sealing & Smart Access
Isolation Room Doors: Pressure, Sealing & Smart Access -
 Hospital Sliding Doors for Treatment Rooms and Clinical Areas
Hospital Sliding Doors for Treatment Rooms and Clinical Areas -
 ICU Doors: What Matters for Safety, Visibility and Infection Control
ICU Doors: What Matters for Safety, Visibility and Infection Control -
 How Airtight Doors Save Building Energy in 2026
How Airtight Doors Save Building Energy in 2026 -
 Do Commercial Buildings Need Fire Rated Doors
Do Commercial Buildings Need Fire Rated Doors -
 7 Maintenance Tips to Extend Automatic Sliding Door Life
7 Maintenance Tips to Extend Automatic Sliding Door Life -
 5 Hospital Door Mistakes That Cost Facilities $50K/Year
5 Hospital Door Mistakes That Cost Facilities $50K/Year -
 Cleanroom Door Installation: Why Seals Fail & How to Fix
Cleanroom Door Installation: Why Seals Fail & How to Fix -
 5 Key Features That Make a Hermetic Door Truly "Hermetic"
5 Key Features That Make a Hermetic Door Truly "Hermetic"

Guangzhou Yizhong Aluminum Industry Co., Ltd.
We are always providing our customers with reliable products and considerate services.
We are always providing our customers with reliable products and considerate services.







Speak Your Mind