
Why Project Managers Trust This Cleanroom Door Manufacturer
- By:Lisa
- 2026-04-23
- 29
Complex infrastructure projects present unique engineering hurdles. Pharma plant construction and hospital operating theater retrofits demand strict planning. Consequently, project managers face three core challenges. First, compliance risk remains non-negotiable. Second, schedule pressure tests every critical milestone. Third, total lifecycle cost requires careful balancing. A cleanroom door serves as the primary physical barrier. Therefore, its quality directly impacts airtightness and microbial control. Furthermore, it dictates long-term maintenance complexity. But why do infrastructure specialists trust a single cleanroom door manufacturer? This article explores four key dimensions. Technical compliance establishes the baseline. Next, product matrices address specific scenarios. Engineering delivery ensures timeline certainty. Finally, supply chain management optimizes budgets. Together, these factors create a reliable assurance system.

I. Compliance First: International Standards & Certification Systems Establish the Project Baseline
Compliance is not optional in pharmaceutical and healthcare projects. During procurement, managers prioritize documentation. Specifically, suppliers must provide auditable technical records.
1.1 Standard Alignment & Pressure Differential Control Logic
Doors must meet strict global standards. For example, ISO 14644-1:2025 and EU GMP Annex 1 apply directly. Additionally, national building codes set baseline requirements. Sterile filling suites demand extreme precision. Consequently, static air leakage must stay below 0.5 m³/h·m at 50 Pa. Professional manufacturers solve this with dual-stage EPDM gaskets. They also integrate automatic drop-down thresholds. As a result, pressure differentials remain stable between 10–15 Pa. This configuration effectively blocks cross-contamination. Conversely, negative-pressure isolation wards face different demands. They require bidirectional resistance from -30 Pa to +30 Pa. Therefore, frame-to-wall junctions need dual sealing. Polyurethane foam and structural adhesive prevent airflow turbulence.
1.2 Dual Endorsement: Fire Safety & Hospital Infection Control Compliance
Hospital projects mandate strict fire ratings. Typically, assemblies require 60–90 minutes of resistance. EN 13501-2 certification proves compliance. Meanwhile, infection control requires durable surfaces. Doors face frequent chemical disinfection daily. For instance, vaporized hydrogen peroxide and 75% ethanol are standard. Consequently, surfaces must pass ISO 22196:2011 testing. Antimicrobial efficacy must exceed 99.9%. Suppliers should provide third-party reports from SGS or TÜV. These documents cover heavy metal leaching and VOC emissions. Ultimately, they integrate into BIM databases. This creates a clear evidentiary chain for FDA audits.
II. Technology-Driven: Vertical Scenario Adaptation & Core Performance Matrix
Project managers seek optimal performance, not just basic compliance. Specialized manufacturers convert field data into precise engineering specs.
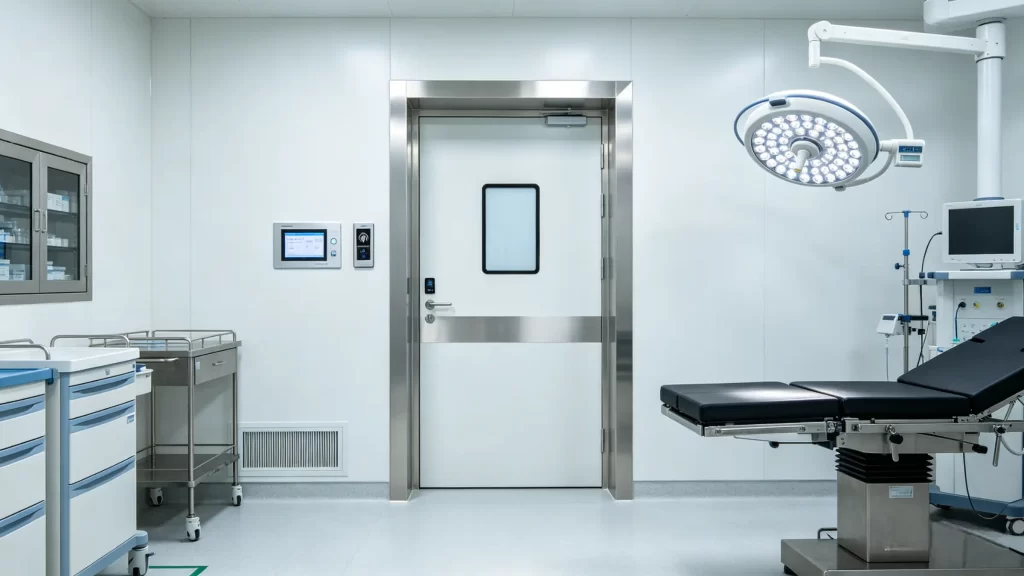
2.1 Material Engineering & Surface Finishes
Different zones require specific material solutions. The following table outlines optimal pairings:
| Application Scenario | Recommended Material Solution | Core Technical Metrics | Project Value |
|---|---|---|---|
| Sterile filling/bioreactor areas | stainless steel cleanroom door (316L) | Surface roughness Ra≤0.4μm, electropolished joints | Eliminates microbial adhesion; supports CIP/SIP validation |
| Operating theaters/ICUs | antibacterial cleanroom door (HPL/aluminum honeycomb) | Silver-ion coating, scratch resistance ≥3H | Reduces HAI rates; extends refurbishment cycles |
| QC labs/chemical testing rooms | laboratory cleanroom door (galvanized steel + powder coat) | Acid/alkali resistant coating, impact resistance ≥15J | Withstands chemical splashes and heavy traffic |
| Imaging/nuclear medicine departments | Lead-equivalent ≥2.0 mmPb radiation shielding door | Seamless rolled lead sheet, equivalent vision panel | Complies with radiation protection standards |
Sterile zones demand precision. Therefore, electropolished joints prevent bacterial adhesion. Operating theaters prioritize impact resistance. Consequently, HPL panels excel in high-traffic wards. QC laboratories face harsh chemicals daily. Thus, powder-coated steel handles acid splashes reliably. Imaging departments require radiation shielding. Specifically, lead-equivalent doors protect staff during scans. For operating room door installations, emergency egress matters most. Heavy-duty pivot hinges ensure smooth operation. Consequently, push forces stay below 80 N during evacuations.
2.2 Opening Mechanisms & Smart Integration
Traditional swing door setups still exist. However, modern facilities prefer automation. automatic sliding door systems now dominate new builds. They reduce particle dispersion effectively. Furthermore, they integrate with Building Management Systems. Pressure differential linkage adjusts airflow instantly. Consequently, pressure recovers within three seconds. Interlock control prevents airlock collapse. PLC logic handles dual-door sequencing seamlessly. Data traceability also improves significantly. RFID modules log every access event. This meets FDA 21 CFR Part 11 requirements. Meanwhile, a cleanroom door with window utilizes double-glazed glass. Argon fills prevent condensation completely. Built-in blinds allow easy inspection. Therefore, structural integrity remains uncompromised.
III. Delivery Control: Full-Lifecycle Project Management & Modular Engineering
Delivery certainty often outweighs raw specifications. Leading manufacturers now rely on digital ecosystems.

3.1 BIM Collaboration & Parametric Prefabrication
Detailed design requires precise coordination. Manufacturers provide LOD 300 Revit libraries first. This enables clash detection with MEP systems. Consequently, tolerances reach ±1.5 mm accuracy. Factory prefabrication handles most assembly work. Hinges, gaskets, and seals arrive pre-installed. As a result, on-site labor drops dramatically. Installation becomes a simple three-step process. Position, secure, and commission. Thus, cleanroom door installation timelines shrink by 40%. Critical path schedules stay protected.
3.2 On-Site Adaptation Logic of modular cleanroom door Systems
Hospital retrofits present unique challenges. Modular frame systems solve tolerance issues easily. They accommodate ±50 mm wall deviations. Quick-release hinges enable rapid maintenance. Consequently, single-door removal takes under 15 minutes. Factory color matching ensures visual consistency. Meanwhile, cleanroom door accessories use standardized interfaces. Floor springs and dust brushes fit perfectly. This eliminates multi-vendor compatibility disputes.
3.3 Logistics Tracking & On-Site Technical Enablement
Large projects require synchronized deliveries. Account managers track every unit via ERP and GPS. Therefore, transit delays become predictable. For international deployments, field engineers provide direct support. They handle torque calibration and smoke tests. Subsequently, operator training ensures proper usage. This approach guarantees a 98% first-pass success rate for automatic cleanroom door systems.
IV. Supply Chain Advantage: Factory-Direct Sourcing & Optimized Decision Costs
Procurement strategy directly impacts project profitability. Partnering with a specialized cleanroom door manufacturer removes unnecessary costs.
4.1 Scaled Production & Cost Transparency
The cleanroom door factory direct model cuts distributor markups. Consequently, pricing aligns closely with BOM costs. Automated production lines accelerate manufacturing cycles. Single-unit completion takes just 7–10 days. Furthermore, framework agreements lock in favorable rates. This protects managers from raw material price spikes.
4.2 Agile Customization & Rapid Response
Pharmaceutical processes evolve rapidly. Custom solutions are now standard practice. Factories support non-standard penetrations easily. Medical gas ports and irregular cutouts require minimal lead time. Additionally, cloud portals offer real-time tracking. Managers view 3D renderings and QC reports instantly. Therefore, project visibility improves dramatically.
4.3 Quantitative Evaluation Criteria for Vendor Selection
Evaluating cleanroom door suppliers requires a weighted approach. Compliance and certification carry the most weight (30%). Technical performance follows closely at 25%. Delivery capability accounts for 20%. After-sales support represents 15%. Finally, commercial terms make up 10%. This scoring model ensures balanced decision-making.
V. Real-Project Validation & Strategic Recommendations
Real-world results validate these engineering approaches.
Case A: mRNA Vaccine Production Facility (East China, 2024)
The client required 180 units for ISO Class 7 zones. They needed full VHP validation and EMA audit readiness. Consequently, the supplier specified 316L stainless steel. Dual-stage FKM seals handled extreme decontamination cycles. Airtightness sampling occurred before shipment. As a result, the facility passed dynamic particle testing immediately. Pressure fluctuations stayed within ±1 Pa. Furthermore, 12-month operational data revealed 24% TCO savings.
Case B: Negative-Pressure Ward Retrofit (South China, 2025)
Emergency delivery demanded 65 sealed door units within 48 hours. Compatibility with existing partitions was mandatory. Therefore, a modular frame system proved essential. Quick-release hinges enabled rapid deployment. Zero on-site cutting occurred. Consequently, installation finished in just 38 hours. Airtightness testing achieved a 100% pass rate. Ultimately, health commission inspections passed without delays.
Project Manager Decision Checklist
- Does the supplier provide a complete gmp cleanroom door technical whitepaper?
- Are third-party CNAS or SGS test reports readily available?
- Do BIM libraries support seamless design coordination?
- Does the modular cleanroom door system tolerate ±50 mm deviations?
- Is full engineering support included during cleanroom door installation?
- Does the cleanroom door factory direct model guarantee scalable production?
Conclusion: The Right Partner Enables Projects to "Get It Right the First Time"
Cleanroom doors are critical engineering nodes. They directly impact compliance and audit outcomes. Consequently, partner selection requires careful evaluation. Tightening regulations demand precise solutions. Compressed schedules leave no room for error. Therefore, working with an experienced cleanroom door manufacturer provides certainty. Every unit must carry international certification. Modular engineering must simplify installation. Full-lifecycle support must reduce long-term costs. When these elements align, trial-and-error ends. Ultimately, controlled environment construction becomes a precise, coordinated process. Project managers can deliver with confidence.
-
 Cleanroom Glass Windows Are The Key to Maintaining a Clean Environment
Cleanroom Glass Windows Are The Key to Maintaining a Clean Environment -
 Top Aluminium Profile Manufacturers in China: Leading the Global Market
Top Aluminium Profile Manufacturers in China: Leading the Global Market -
 The Evolution of Air Tight Sliding Doors
The Evolution of Air Tight Sliding Doors -
 AHU Aluminium Profile: A Comprehensive Guide
AHU Aluminium Profile: A Comprehensive Guide -
 The Importance of Choosing the Right Cleanroom Door in Vietnam
The Importance of Choosing the Right Cleanroom Door in Vietnam -
 The Benefits of Hospital Automatic Doors: Enhancing Efficiency and Safety
The Benefits of Hospital Automatic Doors: Enhancing Efficiency and Safety -
.jpg) The Best Bathroom Door Manufacturers - Unlocking Endless Possibilities!
The Best Bathroom Door Manufacturers - Unlocking Endless Possibilities! -
 Unlock the Possibilities with AJ Manufacturing Doors
Unlock the Possibilities with AJ Manufacturing Doors -
 Make a Statement with Manufactured Home Interior Doors!
Make a Statement with Manufactured Home Interior Doors! -
 what is aluminum profile? Aluminum Profiles for Your Home is the best option
what is aluminum profile? Aluminum Profiles for Your Home is the best option
-
 Why Project Managers Trust This Cleanroom Door Manufacturer
Why Project Managers Trust This Cleanroom Door Manufacturer -
 Aluminum Frame Cleanroom Door: Lab/Hospital Selection Guide
Aluminum Frame Cleanroom Door: Lab/Hospital Selection Guide -
 GMP Cleanroom Door 2026 Compliance Checklist for Pharma
GMP Cleanroom Door 2026 Compliance Checklist for Pharma -
 Door Seal Selection for Cleanroom and Medical Doors
Door Seal Selection for Cleanroom and Medical Doors -
 Aluminum Profile Supplier Selection Checklist
Aluminum Profile Supplier Selection Checklist -
 E-ZONG Ceiling Diffusers for ASHRAE 62.1 Cleanroom Applications
E-ZONG Ceiling Diffusers for ASHRAE 62.1 Cleanroom Applications -
 Avoid 5 Cleanroom Accessories Faults in Food & Beverage Production
Avoid 5 Cleanroom Accessories Faults in Food & Beverage Production -
 E-ZONG Aluminum Diffusers: ASHRAE 170 Ready
E-ZONG Aluminum Diffusers: ASHRAE 170 Ready -
 Manual vs Automatic Cleanroom Doors: Cost & Compliance Guide
Manual vs Automatic Cleanroom Doors: Cost & Compliance Guide -
 E-ZONG Airtight Doors for Contamination-Free Food Plants
E-ZONG Airtight Doors for Contamination-Free Food Plants

Guangzhou Yizhong Aluminum Industry Co., Ltd.
We are always providing our customers with reliable products and considerate services.
We are always providing our customers with reliable products and considerate services.







Speak Your Mind